 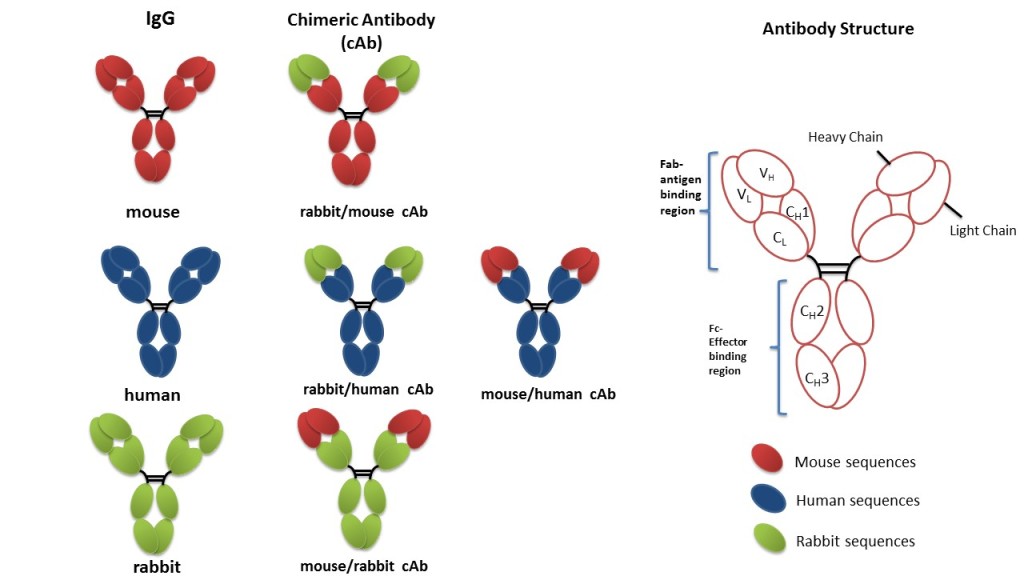
Recombinant antibodies are produced through various genetic engineering / recombinant approaches, known as display technologies, such as: phage display, yeast display, and mammalian cell display. Ig, scFc, Fab, etc.) which enables researchers to use the most suitable form for their application and recombinant antibodies, like classical monoclonal antibodies, can be used in all research applications including immunohistochemistry, immunofluorescence, immunocytochemistry, western blotting, and flow cytometry as well as in diagnostic and therapeutic applications. Importantly, recombinant technologies permit the generation of different antibody forms (i.e. 
For example, Fab can be used to stabilize proteins in structural biology applications and scFv fragments can be used in the screening on the CAR in Car-T therapy or in microscopic applications due to their small size. Each of these formats can have different utilities and can be used in several fields of research as well as in therapeutic applications. Other miniaturized forms of recombinant antibodies include fragment antigen-binding (Fab), which is the antigen-binding region on an antibody composed of two sets of variable and constant components, and multimeric formats like diabodies (dimeric scFvs) or triabodies (trimeric scFvs). Importantly, scFv fragments maintain the specificity of the entire monoclonal antibody and are very easy to generate in a prokaryotic expression system because they are rarely glycosylated. scFv is a fusion protein obtained by recombining the light chains (VL) and heavy chains (VH) of immunoglobulins through a short peptide linker. scFv is the smallest form of a recombinant antibody that is still able to bind the antigen and, despite the removal of the constant regions, retains the specificity of the original Ig. Single-chain fragment variable (scFv) is an antibody fragment composed only of the antibody’s binding site. Recombinant antibodies can come in different formats, including: full-length immunoglobulins (Ig), monovalent antibody fragments such as single-chain fragment variable (scFv) and fragment antigen-binding (Fab), and multimeric formats such as diabodies (dimeric scFvs) or triabodies (trimeric scFvs). These vectors are then introduced into expression hosts such as bacteria, yeast, or mammalian cells for the production of the functional antibody. In recombinant DNA technology, antibody genes encoding for heavy and light chain immunoglobulin fragments are amplified from antibody-producing cells, or synthesized artificially, and cloned into phage vectors. These synthetic antibodies can be used in all applications and have several advantages compared to traditional animal-derived antibodies due to their high specificity, sensitivity, and reproducibility. Recombinant antibodies are generated outside the immune system using synthetic genes and therefore do not require animal immunization for their production. A recombinant antibody is a synthetic monoclonal antibody that is generated by recombinant DNA technology an in vitro alternative to the traditional hybridoma-based technology for creating monoclonal antibodies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
